Cationic ring-opening copolymerization of propylene oxide with tetrahydrofuran by acid exchanged montmorillonite clay | Semantic Scholar
Cationic ring-opening polymerization of a five membered cyclic dithiocarbonate having a tertiary amine moiety - Polymer Chemistry (RSC Publishing)

Anionic Ring-Opening Polymerization of a Functional Epoxide Monomer with an Oxazoline Protecting Group for the Synthesis of Polyethers with Carboxylic Acid Pendants | Macromolecules
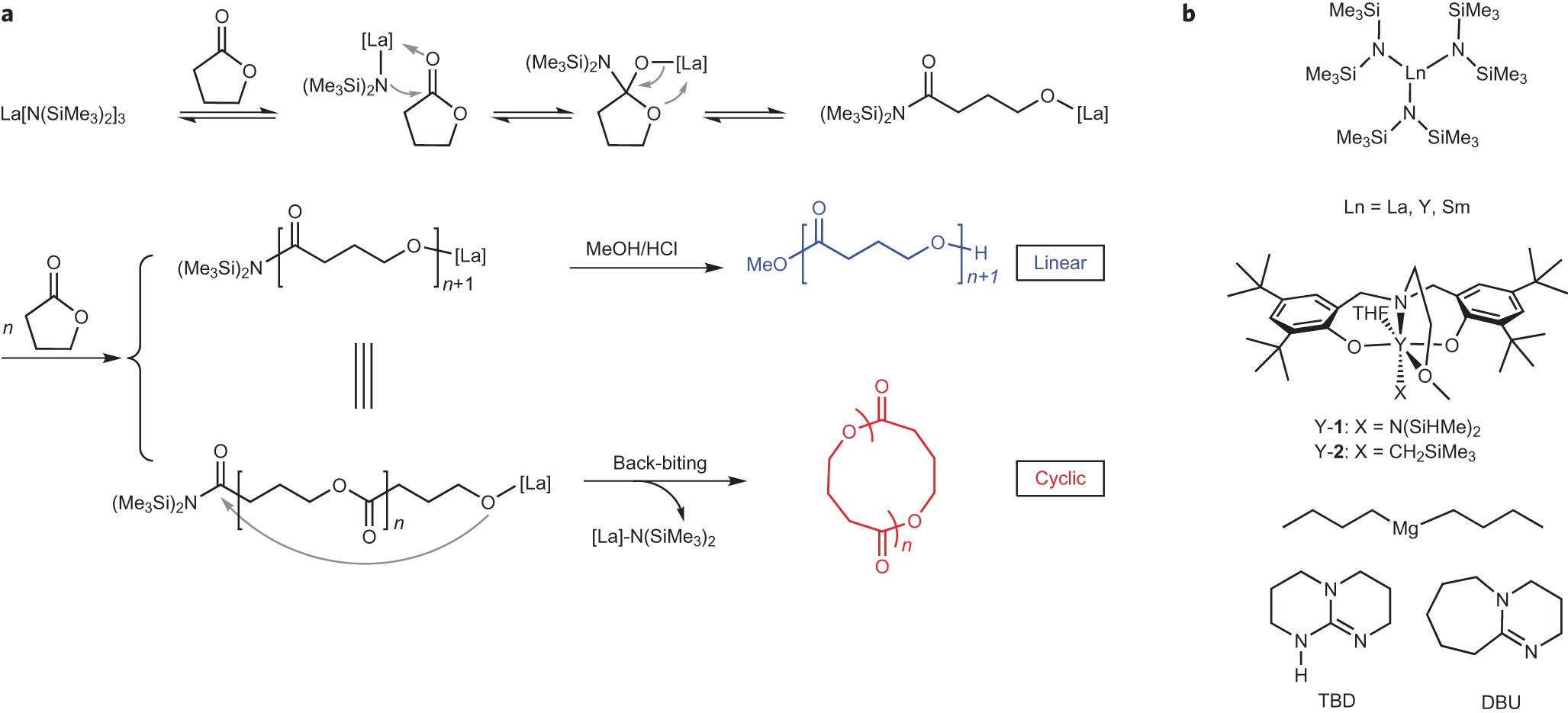
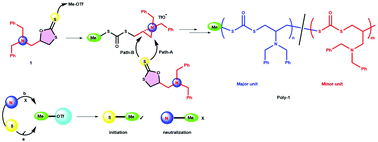
Completely recyclable biopolymers with linear and cyclic topologies via ring -opening polymerization of γ-butyrolactone | Nature Chemistry

Organocatalytic Cationic Ring-Opening Polymerization of a Cyclic Hemiacetal Ester | Industrial & Engineering Chemistry Research

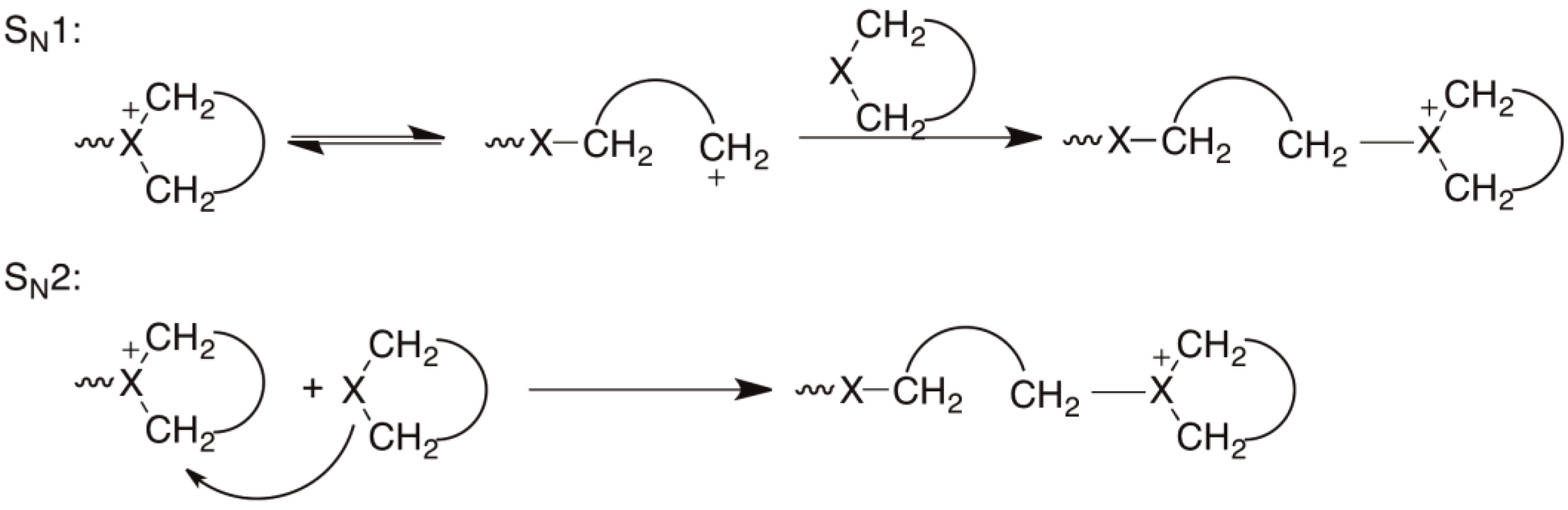
Living Cationic Ring‐Opening Polymerization of Hetero Diels–Alder Adducts to Give Multifactor‐Controlled and Fast‐Photodegradable Vinyl Polymers - Chae - 2023 - Angewandte Chemie International Edition - Wiley Online Library

Chemistry of 2‐oxazolines: A crossing of cationic ring‐opening polymerization and enzymatic ring‐opening polyaddition - Makino - 2010 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

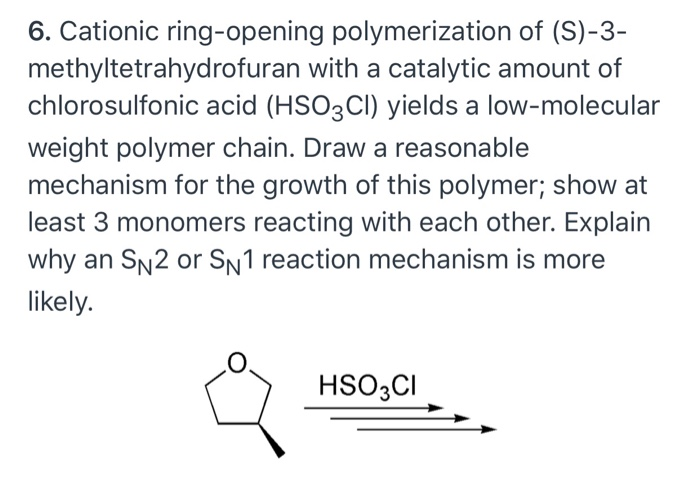
Cationic ring‐opening polymerization (CROP) major mechanistic phenomena - Penczek - 2000 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library

Quasiliving cationic ring-opening polymerization of 2-ethyl-2-oxazoline in benzotrifluoride, as an alternative reaction medium - ScienceDirect

Scheme 13. Mechanism of the cationic ring-opening polymerization of... | Download Scientific Diagram