
RI ) whicho correct. 1 RT (NEET-I 2016) The heat of combustion of carbon to co, e_393.5 kJ/mol. The heat released upon formation of 35.2 g of CO2 from carbon and oxygen

On combustion carbon forms CO and CO2 The heat of formation of CO2 is - 393.5 kJ at of CO is - 110.5 kJ .The heat combustion of CO is

Enthalpy of combustion of carbon to CO2 is -393.5KJ mol-1. Calculate the heat released upon..... - YouTube
The combustion enthalpies of carbon, hydrogen, and methane are 395.5 kJ mol^ 1, 284.8 kJ mol^ 1 and 890.4 kJ mol^ 1 respectively at 25^0C. The value of s†an dard formation enthalpies
Given that: Enthalpy of combustion of Carbon (graphite) = -393kJmol^-1 Enthalpy of combustion of - Tutorke

1. The heats of combustion of carbon, hydrogen and acetylene are -394K.J, - 286K.J and -1301 K.J respectively. Calculate heat of formation of CH2 1) 621 KJ 3) -227 KJ 2) 454 KJ 4) 227 K.J
The heat of combustion of carbon to CO2 is 393.5Kj/mol. The heat rrleased upon formtion of 35.2 g of CO2 from carbn and oxygem gad is
The combustion enthalpie of carbon hydrogen and methane are -395.5 kJ mol^-1 -284.8 kJ mol^-1 and -890.4 kJ - Sarthaks eConnect | Largest Online Education Community
The heat of combustion of carbon to CO2 is 395.5kJ/mol. The heat released upon formation of 35.2 g of CO2 from carbon and oxygen gas is

Calculate the standard heat of formation of carbon disulphide (l). Given that the standard heats of - YouTube

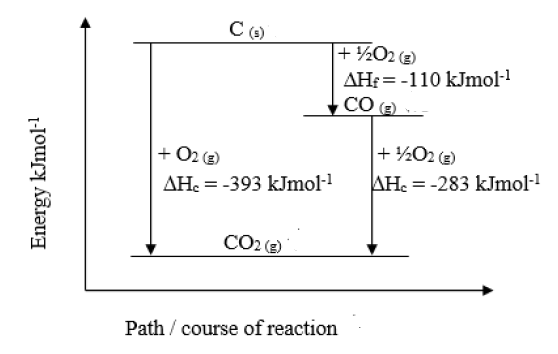
The heat of the combustion of graphite and carbon monoxide respectively are 393.5 kJ mol1 and 283 kJ mol −1. Thus heat of formation of carbon monoxide in kJ mol −1 is:












