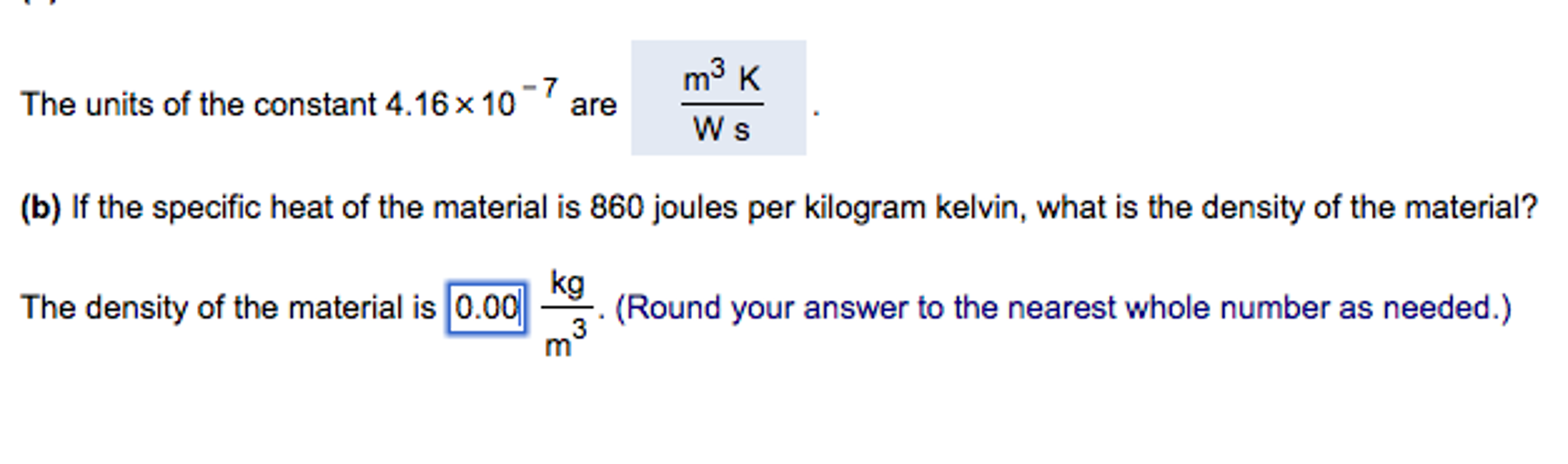
SOLVED: calculate the entropy change in joule/gram kelvein for conversion of 1 gram ice to water at 273 k at 1 ATM pressure if enthalpy of fusion is equals to 6.025 kilo
Specific heat of hydrogen at constant pressure is 30 joule per Kelvin per mol. If unit of length changed to 50 cm, unit of time changes to 1/4 sec and unit of

The enthalpies of formation of n2o and no at 298 Kelvin and 82 and 90 kilo joule per mole the enthalpy of the reaction n2o gas half O2 gives to 2no? - EduRev Class 11 Question
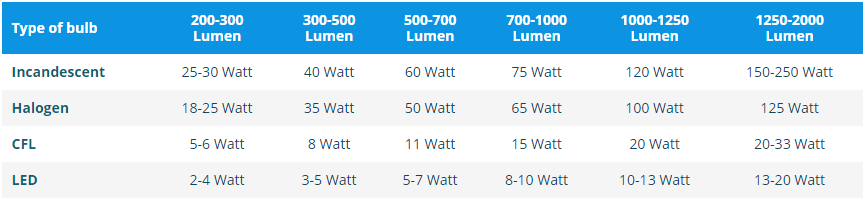
Watt vs. Lumen vs. Kelvin - How to Purchase Lights | SiteLites - Custom Manufacturing & Engineering Inc.

SOLVED: The table provides molar heat capacities (Cm) of selected metals with the units of joules per mole-kelvin. Heat capacity is also frequently given as specific heat capacity (C) with the units