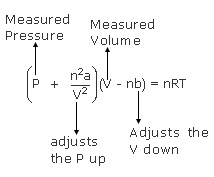
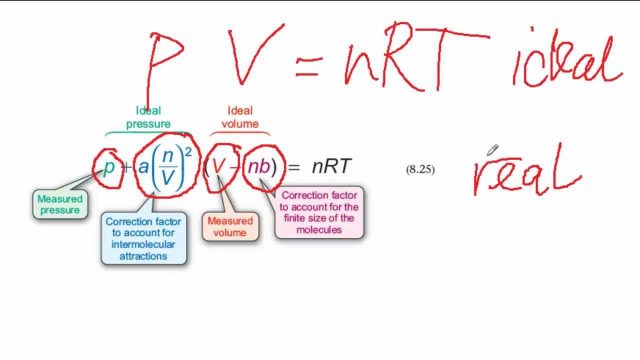
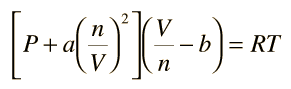The equation of state a van der Waal gas can be expressed as Z=1+dfrac{B}{V_m}+dfrac{C}{V^2_m}+..... If the van der Waal constants a and b are 1.344 litre^2 atm/mol^2 and 0.03 litre mol^{-1}, respectively,
How real gases are different from ideal gases? Derive van der Waal's equation by pressure and volume modifications. - Sarthaks eConnect | Largest Online Education Community